
The results of the interim analysis confirm the ability of idarucizumab to neutralize dabigatran instantaneously, without rebound effect, except in rare patients with very high baseline levels of anticoagulant. Idarucizumab is currently being evaluated in an ongoing Phase III trial, in patients treated with dabigatran presenting with severe active bleeding or requiring emergency surgery or an invasive procedure and are at high risk of bleeding. Specifically, idarucizumab has no anticoagulant or procoagulant effect by itself. In Phase I and II trials, no significant adverse events have been reported. It is rapidly cleared by the kidney with captured dabigatran. Idarucizumab is a humanized fragment of a monoclonal antibody, which binds dabigatran reversibly with high affinity and, when administered intravenously, immediately neutralizes its anticoagulant effect. 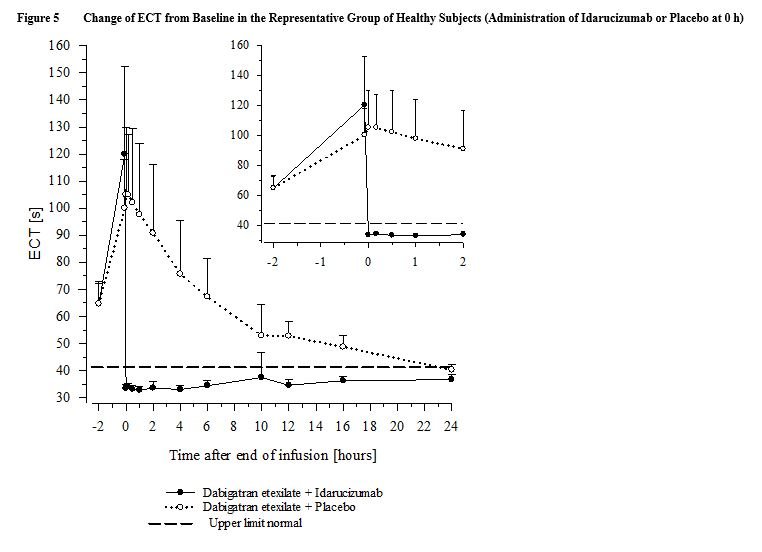 
Hematology Laboratory, Academic Hospital of Toulouse, Hospital Rangueil, Toulouse, FranceĪbstract: Idarucizumab is the first targeted antidote of dabigatran, a direct oral anticoagulant used for prevention and treatment of venous thromboembolism and prevention of stroke in atrial fibrillation.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
